
Batteries – Manufacture of Anodes and Cathodes
With electric cars becoming the “new normal”, wireless consumer electronics making life easier and renewable power systems relying on efficient energy storage, the demand for Lithium Ion batteries is on an exponential growth.
But making these batteries is a complex, costly and quite delicate task!
Read here how RheoStream® can take one major pain out of this complex process.
How To make Batteries
In a battery, anode and cathode are assembled with an electrolyte and a separator, and the “sandwich” is stacked, and formed into the well-known battery cells. Cells are stacked into packs – such as those placed in the bottom of a car.
However, many steps are involved, and each of them pose their own challenges in terms of process design, choice of materials and not the least, quality assurance.
An overview of the process is presented, right, courtesy of “The Battery Report 2023.” Volta Foundation (downloaded April 2, 2024 – https://volta.foundation/battery-report).
The production flow show here starts at the slurry mixing and ends with a battery pack, ready to supply to e.g. a car manufacturer. The capacity is counted in GWh (Giga Watt hours) per year – 1 GWh will provide batteries for about 10-15,000 cars (or millions of cell phones!). Factories making one GWh/year or more is called a “Giga Factory”!

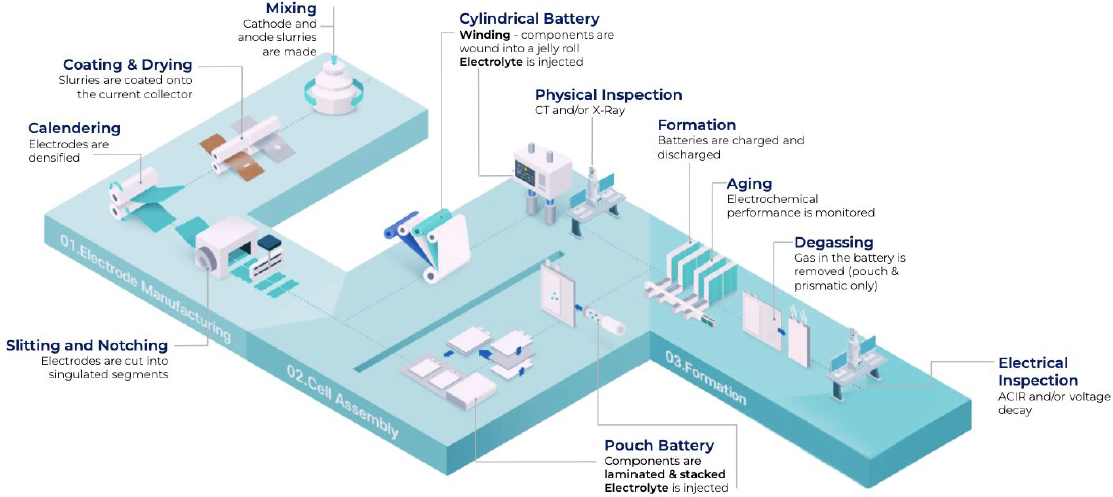
Note: “The Battery Report” can be downloaded for free (link left) and gives a comprehensive overview of the industry and processes.
Why Is Rheology Important in Battery Manufacture?
A battery is not a liquid, so why bother about viscosity?
The first, and very fundamental step, in making batteries is to spread a thin coating of active anode or cathode materials on a thin foil of metal. This way the anode and the cathode are formed. The materials are powders, but to spread a fine powder in a uniform layer only fractions of a millimeter thick the powders are dispersed and mixed with liquids and stabilizers, not unlike making a paint. This “paint” is coated onto the thin metal foil by slot-die coaters.
The coating process may at some gigafactories run at speeds up to 80 m/s coating foils up to 140cm wide – on both sides. The metal is a thin cupper foil, or an aluminum foil (imagine the tinfoil in your kitchen – just thinner). Truly a formidable challenge.
The uniformity of this coating is fundamentally critical to the cycle life, performance, and safety of the final battery pack.
Bottom line: If the viscosity profiles of the anode and cathode slurries are not right, the electrodes will not be usable in the onwards process towards a final battery.

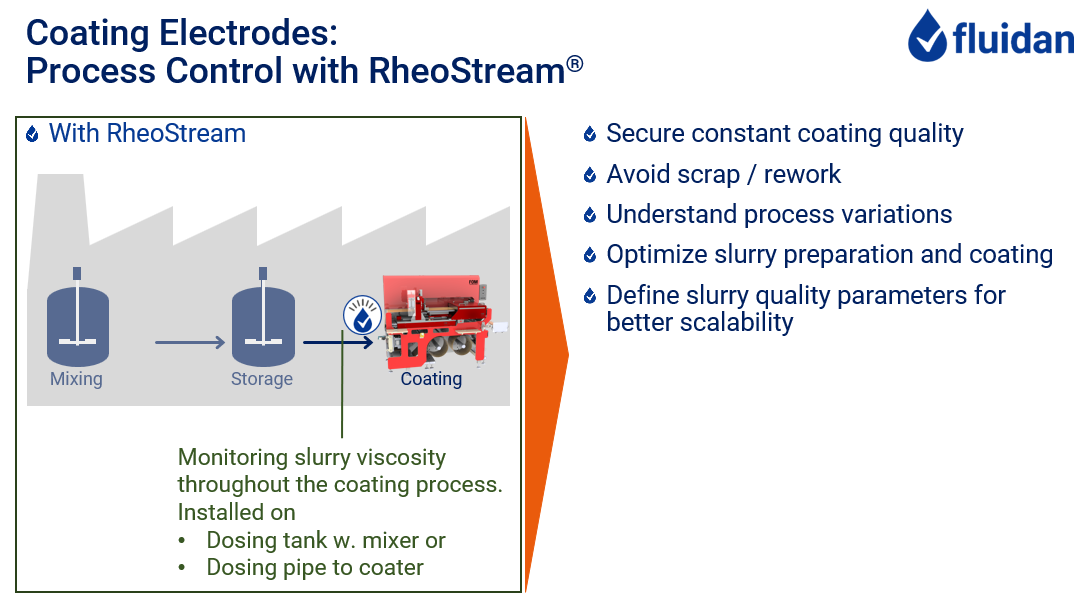
Getting it Right with RheoStream®
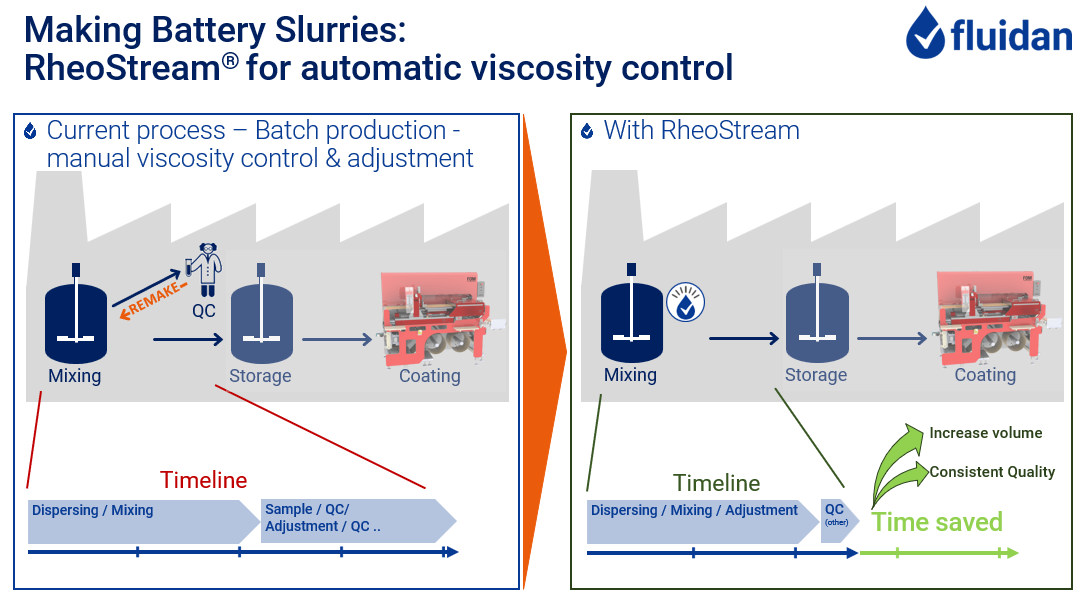
Slurry Mixing:
The anode slurry is typically water-based and is a formulation of graphite, carbon black, CMC (carboxy methyl cellulose – stabilizer) and SBR (styrene butadiene rubber – binder). The slurry is coated onto a cupper foil.
The cathode slurry is normally solvent-borne. It consists of “NMC” (Nickel/Manganese/Cobalt) in NMP (N-methyl pyrrolidone – solvent) with carbon black additive and PVDF binder. The solids content of both could be 50-60%.
The slurries are dispersed and mixed in highly effective mixer vessels.
The most important quality parameter being checked is the viscosity – but the concentration (solids content) is obviously also important.
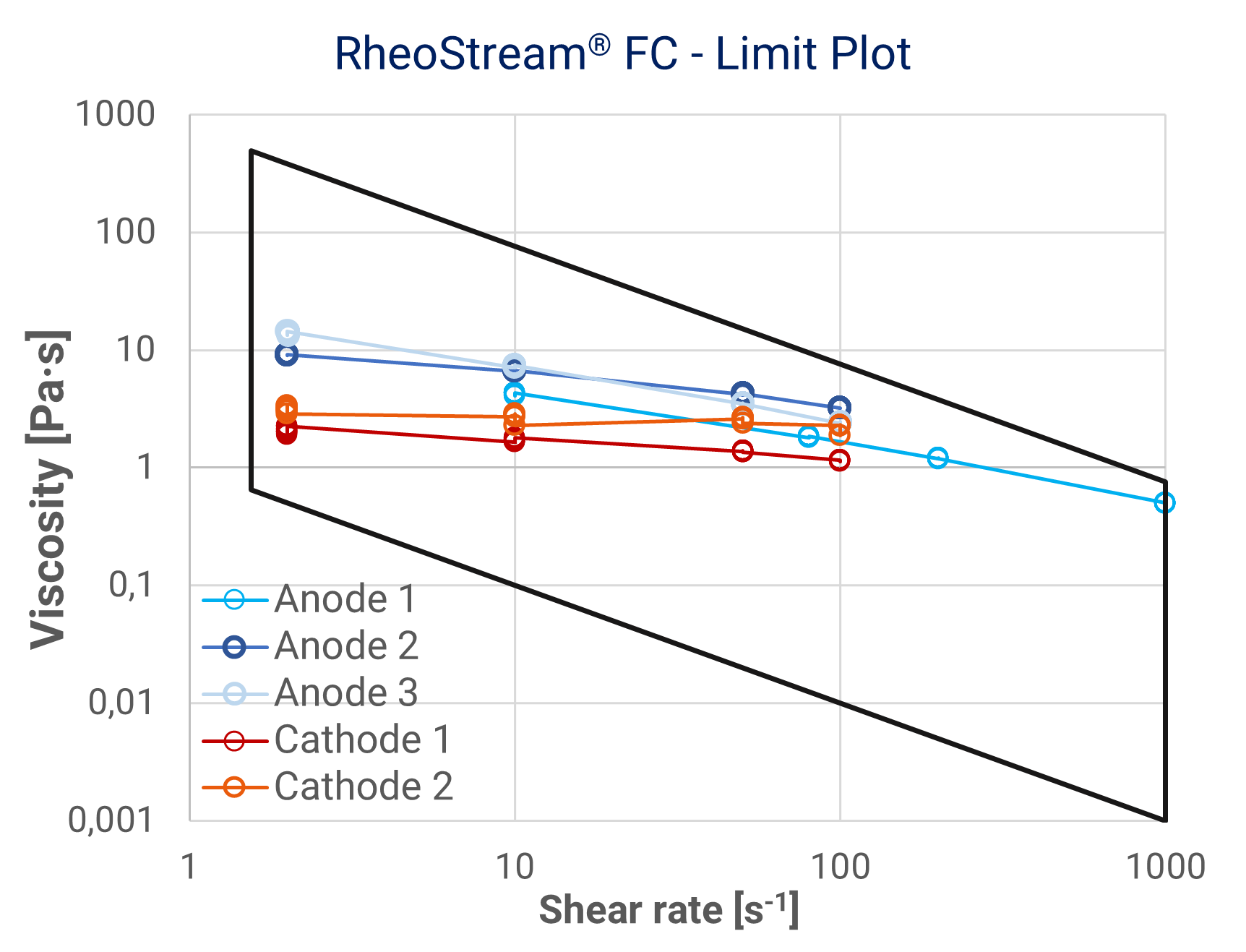
And this is where the trouble starts: The viscosity is not just “a number”. Slurries show a pronounced shear thinning behavior – the viscosity gets lower at high shear.
Some examples are shown, right. Moreover, viscosity is highly temperature dependent. For these reasons, the classic in-line viscometers will not provide reliable measurements. Most battery factories will resort to manual methods, which may provide good quality data – but with a delay and with the associated risk of human error in sample or instrument handling.
RheoStream® is the obvious answer – real-time information about the viscosity curve, makes it possible to adjust the mix and make sure the slurry is right first time, every time.
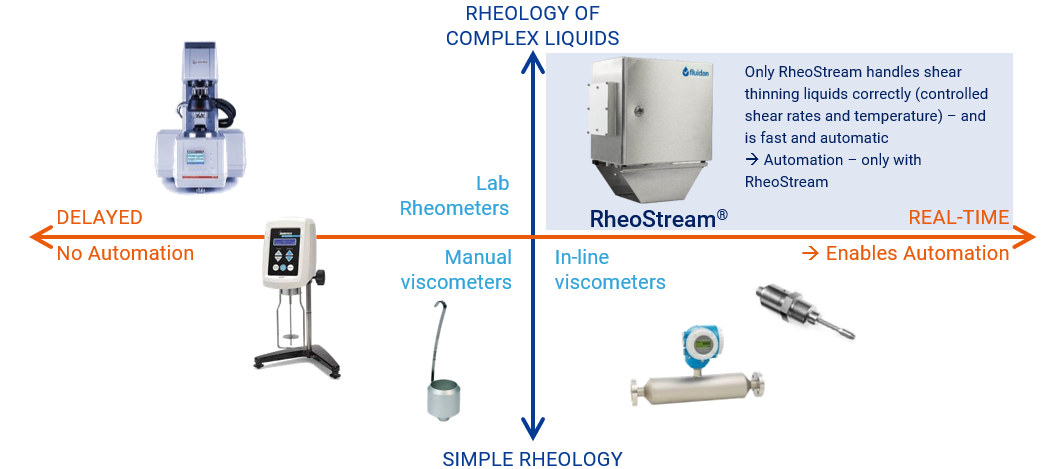
How does it work? RheoStream takes in a sample, adjusts the temperature accurately (e.g. 20°C), and produces a viscosity curve, e.g., 2-1000 s-1. RheoStream exports the values to the factory control system, making it possible to program an automatic correction of the process. With RheoStream, each measurement typically takes 2 minutes, then an updated set of values is ready.
The RheoStream instrument can be mounted directly on the mixing vessel or on a recirculation or bypass.
Imagine never having to wait for the manual analysis and never recycling a batch because it was too thin!
Electrode Coating:
The finished slurries are stored in a buffer tank / day-tank before being pumped to the slot-die coater. Some factories receive the slurry from another location and are not directly in control of the quality.
Either way, if the viscosity is not exactly the same for every batch, or if it is changing over time or if the slurry is not entirely stable, the actual viscosity of the material reaching the coater may be changing from batch to batch or it may be drifting over time.
Then, knowing the viscosity exactly at the time of coating will enable controlling the coating process.
The cathode coating – being solvent based – takes place in a dry room, and the presence of solvent calls for special precautions.
Solvent vapors can be flammable, and explosions can occur. To provide for safe operations RheoStream FCX is recommended. RheoStream FCX is fully solvent compatible and is explosion proof for ATEX zone 1.

Fluidan ApS
Company register: DK36392355
Authorized representatives: Anders L. Østergård (CEO) or Fridolin Okkels (CTO) and Steen Hoff (Chairman of Board)
Privacy
Our privacy policy
Address
Diplomvej 381
2800 Kgs. Lyngby
Denmark
